|
9/22/2023 0 Comments Insulin unit calculator 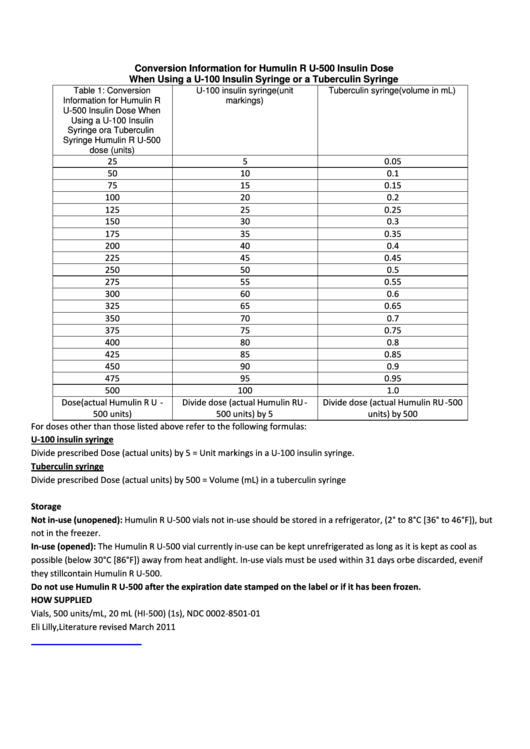
Severe life-threatening, generalized allergy, including anaphylaxis, can occur. Patients should be instructed to always verify the insulin label before each injection. Medication errors, such as accidental mix-ups between basal insulin products and other insulins, particularly rapid-acting insulins, have been reported. Hypoglycemia is the most common adverse reaction of insulin therapy, including Lantus®, and may be life-threatening. Do not administer Lantus® via an insulin pump or intravenously because severe hypoglycemia can occur. If mixed or diluted, the solution may become cloudy, and the onset of action/time to peak effect may be altered in an unpredictable manner. Do not dilute or mix Lantus® with any other insulin or solution. Changes in insulin strength, manufacturer, type, or method of administration may result in the need for a change in insulin dose or an adjustment in concomitant oral antidiabetic treatment. Modify insulin regimen cautiously and only under medical supervision. Monitor blood glucose in all patients treated with insulin. Warnings and Precautions Insulin pens, needles, or syringes must never be shared between patients. Contraindications Lantus® is contraindicated during episodes of hypoglycemia and in patients hypersensitive to insulin glargine or one of its excipients. Limitations of Use: Lantus® is not recommended for the treatment of diabetic ketoacidosis. Lantus® should be administered once a day at the same time every day. Lantus® is a long-acting insulin analog indicated to improve glycemic control in adults and pediatric patients with type 1 diabetes mellitus and in adults with type 2 diabetes mellitus. Increase monitoring with changes to: insulin dose, co-administered glucose lowering medications, meal pattern, physical activity and in patients with hypoglycemia unawareness or renal or hepatic impairment Accidental mix-ups betwe Adjustments in concomitant oral anti-diabetic treatment may be needed Hypoglycemia is the most common adverse reaction of insulin, including Tresiba®, and may be life-threatening. These changes should be made cautiously and under medical supervision. Changes in insulin may affect glycemic control. Sharing poses a risk for transmission of blood-borne pathogens Monitor blood glucose in all patients treated with insulin. Tresiba® is contraindicated during episodes of hypoglycemia and in patients with hypersensitivity to Tresiba® or one of its excipients Never Share a Tresiba® FlexTouch® Pen Between Patients, even if the needle is changed. Tresiba® is not recommended for treating diabetic ketoacidosis or for pediatric patients requiring less than 5 units of Tresiba®. Adjustments in concomitant oral anti-diabetic treatment may be needed Hypoglycemia is the most common adverse reaction of insulin, including Tresiba®, and may be life-threatening Tresiba® (insulin degludec injection) is indicated to improve glycemic control in patients 1 year of age and older with diabetes mellitus.  
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
